|
It is at present impossible to determine whether molecular chaperones are mostly helpers or examiners of mutated proteins because experiments showing either of these roles are very few. This implies that at least some mutations are exposed rather than masked by the activity of molecular chaperones. There is growing evidence that some chaperones, including those dependent on Hsp90, can detect potentially functional but excessively unstable proteins and direct them towards degradation instead of folding. As to why the assistance of mutated proteins by chaperones seems difficult to demonstrate, we note that chaperone-assisted folding can often co-exist with chaperone-assisted degradation. More likely, the insufficient activity of chaperones inflicted a deregulation of multiple cellular systems, including those responsible for signaling and therefore important in development. This effect could not be linked to chaperone-mediated stabilization of mutated proteins. In eukaryotes, it was found that the malfunctioning of chaperones aggravated phenotypic aberrations associated with mutations. These experiments are restricted to bacteria and typically involve overexpression of chaperones. There are some examples that chaperones can alleviate the deleterious effects of mutations through increased assistance of destabilized proteins. We focus on studies describing interactions of chaperones with mutated polypeptides. 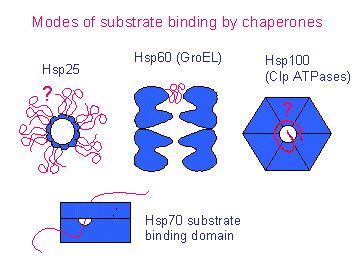
This would allow for the accumulation of genetic variation which could then be exposed during environmental perturbation and facilitate rapid adaptation. It has been proposed that another function of chaperones is to maintain the activity of proteins destabilized by mutation, weakening the selection against suboptimal protein variants. Molecular chaperones help to restore the native states of proteins after their destabilization by external stress.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
